June 17, Skanray Technologies Pvt Ltd. c/o Ms. Yolanda Smith Regulatory Consultant Smith Associates 1468 Harwell Avenue Crofton, Maryland 21114
|
|
|
- Herbert Walton
- 5 years ago
- Views:
Transcription
1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD June 17, 2015 Skanray Technologies Pvt Ltd. c/o Ms. Yolanda Smith Regulatory Consultant Smith Associates 1468 Harwell Avenue Crofton, Maryland Re: Trade/Device Name: Star 90 Regulation Number: 21 CFR Regulation Name: Cardiac Monitor (including Cardiotachometer and Rate Alarm) Regulatory Class: Class II Product Code: MWI Dated: May 12, 2015 Received: May 14, 2015 Dear Ms. Yolanda Smith, We have reviewed your Section 510(k) premarket notification of intent to market the device referenced above and have determined the device is substantially equivalent (for the indications for use stated in the enclosure) to legally marketed predicate devices marketed in interstate commerce prior to May 28, 1976, the enactment date of the Medical Device Amendments, or to devices that have been reclassified in accordance with the provisions of the Federal Food, Drug, and Cosmetic Act (Act) that do not require approval of a premarket approval application (PMA). You may, therefore, market the device, subject to the general controls provisions of the Act. The general controls provisions of the Act include requirements for annual registration, listing of devices, good manufacturing practice, labeling, and prohibitions against misbranding and adulteration. Please note: CDRH does not evaluate information related to contract liability warranties. We remind you, however, that device labeling must be truthful and not misleading. If your device is classified (see above) into either class II (Special Controls) or class III (PMA), it may be subject to additional controls. Existing major regulations affecting your device can be found in the Code of Federal Regulations, Title 21, Parts 800 to 898. In addition, FDA may publish further announcements concerning your device in the Federal Register. Please be advised that FDA's issuance of a substantial equivalence determination does not mean that FDA has made a determination that your device complies with other requirements of the Act or any Federal statutes and regulations administered by other Federal agencies. You must comply with all the Act's requirements, including, but not limited to: registration and listing (21 CFR Part 807); labeling (21 CFR Part 801); medical device reporting (reporting of medical device-
2 Page 2 - Ms. Yolanda Smith related adverse events) (21 CFR 803); good manufacturing practice requirements as set forth in the quality systems (QS) regulation (21 CFR Part 820); and if applicable, the electronic product radiation control provisions (Sections of the Act); 21 CFR If you desire specific advice for your device on our labeling regulation (21 CFR Part 801), please contact the Division of Industry and Consumer Education at its toll-free number (800) or (301) or at its Internet address Also, please note the regulation entitled, "Misbranding by reference to premarket notification" (21 CFR Part ). For questions regarding the reporting of adverse events under the MDR regulation (21 CFR Part 803), please go to for the CDRH's Office of Surveillance and Biometrics/Division of Postmarket Surveillance. You may obtain other general information on your responsibilities under the Act from the Division of Industry and Consumer Education at its toll-free number (800) or (301) or at its Internet address Sincerely yours, Enclosure forbram D. Zuckerman, M.D. Director Division of Cardiovascular Devices Office of Device Evaluation Center for Devices and Radiological Health
3 510(k) Number (if known) Device Name STAR90 DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration Indications for Use Form Approved: OMB No Expiration Date: January See PRA Statement below. Page 1 of 1 Indications for Use (Describe) The STAR 90 multi-parameter Patient Monitoring system is intended to monitor a single adult, pediatric or neonatal patient s vital signs at the bedside or during intra-hospital transport along with the appropriate accessories mentioned / supplied with the unit. Vital signs parameters include ECG (3 lead /5 lead / 12 Lead), SpO2, Respiration, Temperature and Capnography (CO2), IBP. It can also display the digital values of HR/PR, SpO2, RR, Non-Invasive Blood Pressure (Systolic, Diastolic and Mean), Temperature, IBP, EtCO2 and FiCO2 readings. The user, responsible to interpret the monitored data made available, will be a professional health care provider. The device permits patient monitoring with adjustable alarm limits as well as visible and audible alarm signals. The monitor is not intended for home use. Type of Use (Select one or both, as applicable) X Prescription Use (Part21 CFR 801 Subpart D) Over-The-Counter Use (21 CFR 801 Subpart C) PLEASE DO NOT WRITE BELOW THIS LINE- CONTINUE ON A SEPARATE PAGE IF NEEDED. FOR FDA USE ONLY Concurrence of Center for Devices and Radiological Health (CDRH) (Signature) This section applies only to requirements of the Paperw ork Reduction Act of *DO NOT SEND YOUR COMPLETED FORM TO THE PRA STAFF ADDRESS BELOW.* The burden time for this collection of information is estimated to average 79 hours per response, including the time to review instructions. search existing data sources, gather and maintain the data needed and complete and review the collection of information. Send comments regarding this burden estimate or any other aspect of this information collection, including suggestions for reducing this burden, to: Department of Health and Human Services Food and Drug Administration Office of Chief Information Officer Paperw ork Reduction Act (PRA) Staff PRAStaff@fda.hhs.gov "An agency may not conduct or sponsor, and a person is not required to respond to, a collection of information unless it displays a currently valid OMB number." FORM FDA 3881 (1114) Page 1 of 1 PSC Publishmg Services(301} EF 1
4 510(k) Summary (As Per 21 CFR ) Page 1 of 4 SPONSOR Company Name: Skanray Technologies PVt Ltd Healthcare Division Company Address: Skanray Technologies PVT Ltd. No , Hebbal Industrial Area, Mysore, KA India Telephone: Fax: Contact Person: Hema Singh Summary Prepared September 5, 2014 Trade Name: Star 90 Common/Usual Name: Patient Monitoring System Classification Name: Physiological Patient Monitor (without arrhythmia detection or alarms) Product Code: MWI Device Class: Class II Regulation Number: 21 CFR Predicate Device Company Product 510(k) # Larsen & Toubro Ltd Star 55 K08073 Philips medizin Systeme Boeblingen Philips Intellivue patient Monitor K Device Description The STAR 90 Multi Parameter Patient Monitoring System is a Semi Modular Patient monitor with a wide range of communication options, touch screen and capacitive sensing buttons. Modularity in this monitor is achied by providing 3 slots wherein the parameter modules that fit into these modules are hot swappable i.e. they can be connected or disconnected from the unit without switching off the unit. The module is detected at run time. Patient monitoring modules include: ECG+RESPIRATION+TEMPERATURE MODULE: This module is divided in area and functionality between the interface card and the ECG child card. The Temperature section and the DC DC section of the ER2T lie in the interface card.
5 Page 2 of 4 SpO2: This module is inbuilt inside the monitor on the interface card, but there is an option to jack an external SpO2 module in one of the jackable slots. It is used to measure the partial pressure of oxygen in the human body. NIBP: This module is inbuilt inside the monitor on the interface card. It is used to non invasively measure the systolic and diastolic blood pressure. CO2: This module can be jacked into one of the jackable slots. It is used to measure the level of CO2 in the blood (EtCO2 & FiCO2). When AGM is used this module will not be available IBP: STAR 90 can support 2 IBP modules. For ease of understanding we shall name the 1st IBP as IBP ½ and the 2nd IBP as IBP ¾. IBP ½ module is inbuilt in the interface card, whereas the IBP ¾ can be jacked in one of the slots. It is used to invasively measure the systolic and diastolic blood pressure AGM: The connectivity for this module is inbuilt inside the interface card of the system, but the AGM module has to be connected through the connector provided. It is used to measure anesthesia gas concentration, CO2 and O2.This is optional module in place of CO2 module. Indications for Use The STAR 90 Multi Parameter Patient Monitoring System is intended to monitor a single adult, pediatric or neonatal patient s vital signs at the bedside or during intra hospital transport along with the appropriate accessories mentioned / supplied with the unit. Vital signs parameters include ECG (3 lead / 5 lead / 12 lead), Spo2, Respiration, Temperature and Capnography (CO2), IBP. It can also display the numeric values of HR/PR, SpO2, RR, Non Invasive Blood Pressure (Systolic, Diastolic and Mean). Temperature, IBP, EtCO2 and FiCO2 readings. The user, responsible to interpret the monitored data made available, will be a professional health care provider. The device permits patient monitoring with adjustable alarm limits as well as visible and audible alarm signals. The monitor is not intended for home use. Summary of Technological Characteristics Star 90 is designed and developed with reference to our previous 510K cleared product Star55 with same product code as Star90 MWI. These 2 products are same in terms of: ECG, Respiration, Temperature, IBP, NIBP,
6 Page 3 of 4 SpO2 and CO2 Display type Alarm systems Thermal recorder / printer Equipment classification, applicable standards Star90 is compared with MP70 for advanced features that are included in the Star 90 and include: Communication with external equipment including analog and system outputs Display size and resolution. Differences between Star 90 and Star 55 The differences between the two devices includes advanced features that include: Number of waveform traces displayed Waveform display options / selections The inclusion of the advanced features have been validated and verified to ensure that the new features do not impact performance and the safety and efficacy of the device. Non Clinical Testing Electrical and EMC Testing for the Star 90 included the following IEC EMC Test Report IEC Medical electrical equipment Part 1: General requirements for basic safety and essential performance IEC Medical electrical equipment Part 1 6: General requirements for basic safety and essential performance Collateral standard: Usability IEC Medical electrical equipment Part 1 8: General requirements for basic safety and essential performance Collateral standard: General requirements, tests and guidance for alarm systems in medical electrical equipment and medical electrical systems IEC Medical electrical equipment Part 2 27: Particular performance of electrocardiographic monitoring equipment IEC Medical electrical equipment Part 2 30: Particular performance of automatic cycling non invasive blood pressure monitoring equipment.
7 IEC IEC ISO ISO ISO Page 4 of 4 Medical electrical equipment Part 2 34: Particular performance of invasive blood pressure monitoring equipment. Medical electrical equipment Part 2 49: Particular performance of multifunction patient monitoring equipment Medical electrical equipment Part 2 55: Particular performance of respiratory gas monitors Medical electrical equipment Part 2 56: Particular requirements for basic safety and essential performance of clinical thermometers for body temperature measurement Medical electrical equipment Part 2 61: Particular requirements for basic safety and essential performance of pulse Oximeter equipment Additional Performance Testing: ANSI/AAMI EC 13 Cardiac Monitors, Heart rate Meter and Alarms ANSI/SSMI SP10 Manual, Electronic or Automated Sphygmomanometers IEC Medical device software software life cycle processes (Software/Informatics) ISO Application of risk management to medical devices Substantial Equivalence The Star 90 is substantially equivalent to the predicate device in Indications for Use, Materials and Design. Safety and performance testing was performed and Skanray Technologies has concluded that the device does not introduce any significant questions of safety and efficacy and is substantially equivalent to the predicate devices.
February 23, Skanray Technologies Private Limited Yolanda Smith, Consultant Smith Associates 1468 Harwell Ave Crofton, Maryland 21114
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service February 23, 2016 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Skanray
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service February 23, 2016 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Skanray
TOUBRO LIMITED ELECTRICAL & ELECTRONICS DIVISION - ELECTRONIC PRODUCTS 510(K) SUMMARY. (Per section )
 LARSEN & TOUBRO LIMITED SEP 2 1 2O Mysore Complex, KIADB Industrial Area, Hebbal -Hootagalli, Mysore -570 018. Tel : +91 (821) 2402561 Fax: +91(821) 2402468 E -Mail: Ref: 30 th March 2007 Page: 01 of 04
LARSEN & TOUBRO LIMITED SEP 2 1 2O Mysore Complex, KIADB Industrial Area, Hebbal -Hootagalli, Mysore -570 018. Tel : +91 (821) 2402561 Fax: +91(821) 2402468 E -Mail: Ref: 30 th March 2007 Page: 01 of 04
Lc~rlI. z - 510(k) Summary JUL
 JUL - 2 2008 Premarket Notification 510(k) Submission-5 10(k) Summary Report No. Report No. TE20071I01 I Lc~rlI 510(k) Summary z - This summary of 510(k) safety and effectiveness information is being submitted
JUL - 2 2008 Premarket Notification 510(k) Submission-5 10(k) Summary Report No. Report No. TE20071I01 I Lc~rlI 510(k) Summary z - This summary of 510(k) safety and effectiveness information is being submitted
E. GentleLASE Family of Laser Systems 510k Summary JL1
 General Information: E. GentleLASE Family of Laser Systems 510k Summary JL1 This 5 10(k) is to provide notification of substantial equivalence for the Candela GentleLASE Family of Laser Systems, which
General Information: E. GentleLASE Family of Laser Systems 510k Summary JL1 This 5 10(k) is to provide notification of substantial equivalence for the Candela GentleLASE Family of Laser Systems, which
TRUSTED PRECISION. A HISTORY OF INNOVATION. Valleylab FT10 Energy Platform Product Information Kit
 TRUSTED PRECISION. A HISTORY OF INNOVATION. Valleylab FT10 Energy Platform Product Information Kit A HERITAGE YOU CAN TRUST The Valleylab energy portfolio You can have confidence in almost 50 years of
TRUSTED PRECISION. A HISTORY OF INNOVATION. Valleylab FT10 Energy Platform Product Information Kit A HERITAGE YOU CAN TRUST The Valleylab energy portfolio You can have confidence in almost 50 years of
B. Company Address: N. Kendall Dr. Miami, Florida 92121
 VIII. PREMARKET NOTIFICATION 510K SUMMARY SUBMITTER INFORMATION A. Company Name: Drip Alert, Inc. B. Company Address: 13882 N. Kendall Dr. Miami, Florida 92121 C. Company Phone: 305-385-8000 Company Fax:
VIII. PREMARKET NOTIFICATION 510K SUMMARY SUBMITTER INFORMATION A. Company Name: Drip Alert, Inc. B. Company Address: 13882 N. Kendall Dr. Miami, Florida 92121 C. Company Phone: 305-385-8000 Company Fax:
Regulation of Alarm Remote Notification Systems
 Regulation of Alarm Remote Notification Systems Shawn Forrest Center for Devices and Radiological Health Food and Drug Administration Medical Device Alarms Summit October 5, 2011 Evolving Clinical Environment
Regulation of Alarm Remote Notification Systems Shawn Forrest Center for Devices and Radiological Health Food and Drug Administration Medical Device Alarms Summit October 5, 2011 Evolving Clinical Environment
SPECIFICATION FOR MULTIPARAMETER MONITOR
 SPECIFICATION FOR MULTIPARAMETER MONITOR Eight channel high resolution colour TFT display with 12.1" screen size and auto spacing User Selectable Display formats and waveform colours. ECG monitoring -
SPECIFICATION FOR MULTIPARAMETER MONITOR Eight channel high resolution colour TFT display with 12.1" screen size and auto spacing User Selectable Display formats and waveform colours. ECG monitoring -
Omni (K) PATIENT MONITOR
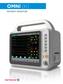 Omni (K) PATIENT MONITOR Omni (K) Intuitive Designed for a fast paced work environment, the Infinium Omni (K) patient monitor offers an extremely simple and adaptable user interface. Patient information
Omni (K) PATIENT MONITOR Omni (K) Intuitive Designed for a fast paced work environment, the Infinium Omni (K) patient monitor offers an extremely simple and adaptable user interface. Patient information
OMNI TOUCH SCREEN PATIENT MONITOR
 OMNI TOUCH SCREEN PATIENT MONITOR OMNI Intuitive Designed for a fast paced work environment, the Infinium Omni patient monitor offers an extremely simple and adaptable user interface. Patient information
OMNI TOUCH SCREEN PATIENT MONITOR OMNI Intuitive Designed for a fast paced work environment, the Infinium Omni patient monitor offers an extremely simple and adaptable user interface. Patient information
PM-9000 Express Portable Patient Monitor
 FEATURES 12.1" color TFT with maximum 8 waveforms Rich in ECG software OxyCRG software available 40 seconds full-disclosure waveforms review Drug dose calculation Bed to bed view Networking capacity Multi-Gas/O2
FEATURES 12.1" color TFT with maximum 8 waveforms Rich in ECG software OxyCRG software available 40 seconds full-disclosure waveforms review Drug dose calculation Bed to bed view Networking capacity Multi-Gas/O2
Ultraview SL2400 With Wireless-LAN-Option
 Ultraview SL2400 With Wireless-LAN-Option Spacelabs Healthcare Connecting Innovation with Care Spacelabs Healthcare is fully committed to making strong connections: between clinicians and their patients;
Ultraview SL2400 With Wireless-LAN-Option Spacelabs Healthcare Connecting Innovation with Care Spacelabs Healthcare is fully committed to making strong connections: between clinicians and their patients;
T1 BEDSIDE MONITOR GUIDELINE
 APPENDIX A T1 BEDSIDE MONITOR GUIDELINE 5.23 Software Version This document is a guideline only to be used as an aid to comprehensive Inservice training. NAME: HOSPITAL: DATE: VALIDATOR: A. OVERVIEW 1.
APPENDIX A T1 BEDSIDE MONITOR GUIDELINE 5.23 Software Version This document is a guideline only to be used as an aid to comprehensive Inservice training. NAME: HOSPITAL: DATE: VALIDATOR: A. OVERVIEW 1.
PATIENT MONITORING SYSTEMS
 PATIENT MONITORING SYSTEMS PATIENT MONITORING SYSTEMS The benefits of HEYER Scalis to meet the market situation HEYER Scalis function & features make it fit to match the requirement of standard applications
PATIENT MONITORING SYSTEMS PATIENT MONITORING SYSTEMS The benefits of HEYER Scalis to meet the market situation HEYER Scalis function & features make it fit to match the requirement of standard applications
CLEO VERSATILITY IN VITAL SIGNS
 CLEO VERSATILITY IN VITAL SIGNS CLEO PORTABLE weighs less than 3 lbs TOUCHSCREEN easy and intuitive to use MOBILE backup battery powered The Cleo is a new and intuitive approach to patient vital signs
CLEO VERSATILITY IN VITAL SIGNS CLEO PORTABLE weighs less than 3 lbs TOUCHSCREEN easy and intuitive to use MOBILE backup battery powered The Cleo is a new and intuitive approach to patient vital signs
5/ 5i/ 5c/ 7/ 7i/ 7c/ 5 elite/ 5i elite/ 5c elite/ 7 elite/ 7i elite/ 7c elite
 Summary The élance 5 / 5i / 5c / 7 / 7i / 7c /5 elite/ 5i elite / 5c elite / 7 elite / 7i elite /7c elite are integrated vital signs monitors with widescreen resistive touchscreen display. The monitors
Summary The élance 5 / 5i / 5c / 7 / 7i / 7c /5 elite/ 5i elite / 5c elite / 7 elite / 7i elite /7c elite are integrated vital signs monitors with widescreen resistive touchscreen display. The monitors
PATIENT MONITORING SYSTEMS
 PATIENT MONITORING SYSTEMS Patient Monitoring Systems DESIGN FEATURES HEYER VizOR is our premium solution for sophisticated patient monitoring applications. VizOR comes in its standard configuration with
PATIENT MONITORING SYSTEMS Patient Monitoring Systems DESIGN FEATURES HEYER VizOR is our premium solution for sophisticated patient monitoring applications. VizOR comes in its standard configuration with
PATIENT MONITORING SYSTEMS
 PATIENT MONITORING SYSTEMS Patient Monitoring Systems DESIGN FEATURES HEYER VizOR is our premium solution for sophisticated patient monitoring applications. VizOR comes in its standard configuration with
PATIENT MONITORING SYSTEMS Patient Monitoring Systems DESIGN FEATURES HEYER VizOR is our premium solution for sophisticated patient monitoring applications. VizOR comes in its standard configuration with
Ultraview SL Command Module Parameter Set ECG NIBP TEMP IP CO SpO2 Options SL Command Module: A B Our goals are simple:
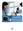 Ultraview SL2700 Spacelabs Healthcare - Connecting Innovation with Care Spacelabs Healthcare is fully committed to making strong connections: between clinicians and their patients; between technological
Ultraview SL2700 Spacelabs Healthcare - Connecting Innovation with Care Spacelabs Healthcare is fully committed to making strong connections: between clinicians and their patients; between technological
OMNI Express VS VERSATILITY IN VITAL SIGNS
 OMNI Express VS VERSATILITY IN VITAL SIGNS OMNI Express VS TOUCHSCREEN easy and intuitive to use PORTABLE weighs less than 6lbs MOBILE backup battery powered The Omni Express VS is a new intuitive approach
OMNI Express VS VERSATILITY IN VITAL SIGNS OMNI Express VS TOUCHSCREEN easy and intuitive to use PORTABLE weighs less than 6lbs MOBILE backup battery powered The Omni Express VS is a new intuitive approach
GE Healthcare. Dash Variable-Acuity Monitoring
 GE Healthcare Dash Variable-Acuity Monitoring The RIGHT information. Right now. In your busy schedule, having accurate patient information at your fingertips is critical. And there is no time for false
GE Healthcare Dash Variable-Acuity Monitoring The RIGHT information. Right now. In your busy schedule, having accurate patient information at your fingertips is critical. And there is no time for false
MRI Patient Monitoring System (Model )
 MRI Patient Monitoring System (Model 865214) INSTRUCTIONS FOR USE Revision C English A Specifications Specifications PATIENT SAFETY GENERAL Conforms to UL STD 60601-1. Certified to CAN/CSA STD C22.2 No.
MRI Patient Monitoring System (Model 865214) INSTRUCTIONS FOR USE Revision C English A Specifications Specifications PATIENT SAFETY GENERAL Conforms to UL STD 60601-1. Certified to CAN/CSA STD C22.2 No.
General Skills. yes. Device controls, buttons, indicators, and connectors
 HeartStart MRx Skills Checklist This checklist assesses qualified medical personnel s understanding of MRx operation after instructor-based training. Perform a quick skills assessment using the General
HeartStart MRx Skills Checklist This checklist assesses qualified medical personnel s understanding of MRx operation after instructor-based training. Perform a quick skills assessment using the General
The Blood Pressure Monitor is digital upper arm monitors intended for use in measuring blood pressure and heartbeat rate with upper arm circumference
 The Blood Pressure Monitor is digital upper arm monitors intended for use in measuring blood pressure and heartbeat rate with upper arm circumference ranging from 22cm to 32 cm ( about 83/4-121/2 ).It
The Blood Pressure Monitor is digital upper arm monitors intended for use in measuring blood pressure and heartbeat rate with upper arm circumference ranging from 22cm to 32 cm ( about 83/4-121/2 ).It
DPM Summary of Features and Benefits
 Summary of Features and Benefits PAT I E N T M O N I TO R 12.1" TFT display, configurable up to 8 waveforms, enables maximum data analysis of your most critical parameters Standard features include 3/5-lead
Summary of Features and Benefits PAT I E N T M O N I TO R 12.1" TFT display, configurable up to 8 waveforms, enables maximum data analysis of your most critical parameters Standard features include 3/5-lead
Ensuring the Effectiveness of Clinical Alarm Systems
 Ensuring the Effectiveness of Clinical Alarm Systems Requirements: Ensure that critical alarms used in the clinical environment are tested on an ongoing, scheduled basis and they are sufficiently audible
Ensuring the Effectiveness of Clinical Alarm Systems Requirements: Ensure that critical alarms used in the clinical environment are tested on an ongoing, scheduled basis and they are sufficiently audible
Passport 12m and Passport 17m BEDSIDE MONITOR GUIDELINE 5.21 Software Version NAME: HOSPITAL: DATE: VALIDATOR: Performed
 Passport 12m and Passport 17m BEDSIDE MONITOR GUIDELINE 5.21 Software Version This document is a guideline only to be used as an aid to comprehensive Inservice training. NAME: HOSPITAL: DATE: VALIDATOR:
Passport 12m and Passport 17m BEDSIDE MONITOR GUIDELINE 5.21 Software Version This document is a guideline only to be used as an aid to comprehensive Inservice training. NAME: HOSPITAL: DATE: VALIDATOR:
qube mini Overview
 D a t a S h e e t qube mini Overview The 91389 qube mini is a compact, robust, and light-weight patient monitor with extensive clinical usability. Key features of the qube mini monitor include: Eye-level
D a t a S h e e t qube mini Overview The 91389 qube mini is a compact, robust, and light-weight patient monitor with extensive clinical usability. Key features of the qube mini monitor include: Eye-level
B40 Patient Monitor. Affordable clinical excellence. Advanced clinical parameters. Ease of use for fast decision-making. Performance and reliability
 B40 Patient Monitor Affordable clinical excellence Patients with acute, life-threatening conditions need the best possible care. The B40 Monitor from GE Healthcare provides a continuous flow of quality
B40 Patient Monitor Affordable clinical excellence Patients with acute, life-threatening conditions need the best possible care. The B40 Monitor from GE Healthcare provides a continuous flow of quality
Multi-Parameter Patient Monitor
 KIZLON Multi-Parameter Patient Monitor Email: info@kizlonmedical.com Website: www.kizlonmedical.com Multi-Parameter Patient Monitor KPM-A1 Series s of Portable Patient Monitor KPM-A1 Series has color TFT
KIZLON Multi-Parameter Patient Monitor Email: info@kizlonmedical.com Website: www.kizlonmedical.com Multi-Parameter Patient Monitor KPM-A1 Series s of Portable Patient Monitor KPM-A1 Series has color TFT
GE Healthcare Healthcare Systems 9900 Innovation Drive Wauwatosa, WI USA
 URGENT Field Safety Notice GE Healthcare Healthcare Systems 9900 Innovation Drive Wauwatosa, WI 53226 USA 22 nd January 2014 GEHC Ref 36092 To: Healthcare Administrator / Risk Manager Chief of Nursing
URGENT Field Safety Notice GE Healthcare Healthcare Systems 9900 Innovation Drive Wauwatosa, WI 53226 USA 22 nd January 2014 GEHC Ref 36092 To: Healthcare Administrator / Risk Manager Chief of Nursing
QAM.L L550 series. Patient Monitor Manual QAM.L
 QAM.L550.1112.2 L550 series Patient Monitor Manual QAM.L550.0915.3 Preface This manual has been provided to give detailed descriptions on DARAY s L550 patient monitor, covering the performance, operation
QAM.L550.1112.2 L550 series Patient Monitor Manual QAM.L550.0915.3 Preface This manual has been provided to give detailed descriptions on DARAY s L550 patient monitor, covering the performance, operation
MRI Monitor. All-in-one monitor for the MRI environment. Features. Parameters. Accessories
 MRI Monitor All-in-one monitor for the MRI environment The MRI Monitor helps manage Clinical Information Logistics needs by enabling caregivers to access important information at the point of care. Designed
MRI Monitor All-in-one monitor for the MRI environment The MRI Monitor helps manage Clinical Information Logistics needs by enabling caregivers to access important information at the point of care. Designed
GE Healthcare. Quick Guide. Dash 2500 Patient Monitor
 GE Healthcare Quick Guide Dash 2500 Patient Monitor Hardkeys Main Menu Hide menu and view entire waveform screen Display the large numeric vital signs Review or change alarm limits for all operating parameters
GE Healthcare Quick Guide Dash 2500 Patient Monitor Hardkeys Main Menu Hide menu and view entire waveform screen Display the large numeric vital signs Review or change alarm limits for all operating parameters
Vitalmax 4000 CL Product Specifications
 Page 1 of 5 Vitalmax 4000 CL 510 Garden Avenue North, Clearwater, FL 33755 Phone: (727) 442-8118 Fax: (727) 443-7257 A. Mechanical Description Size 10" H x 13" W x 8"D (25 cm x 33 cm x 20 cm) Weight 18
Page 1 of 5 Vitalmax 4000 CL 510 Garden Avenue North, Clearwater, FL 33755 Phone: (727) 442-8118 Fax: (727) 443-7257 A. Mechanical Description Size 10" H x 13" W x 8"D (25 cm x 33 cm x 20 cm) Weight 18
Nellcor Bedside SpO 2 Patient Monitoring System, PM100N
 Nellcor Bedside SpO 2 Patient Monitoring System, PM100N In-Service Presentation Nellcor Bedside SpO 2 Patient Monitoring System is CE marked and commercially av ailable in European Union countries. Nellcor
Nellcor Bedside SpO 2 Patient Monitoring System, PM100N In-Service Presentation Nellcor Bedside SpO 2 Patient Monitoring System is CE marked and commercially av ailable in European Union countries. Nellcor
HeartStart XL+ Configuration Worksheet
 HeartStart XL+ Configuration Worksheet The following pages show the available configuration options for the HeartStart XL+. Please enter your configuration choices and save this worksheet for your records.
HeartStart XL+ Configuration Worksheet The following pages show the available configuration options for the HeartStart XL+. Please enter your configuration choices and save this worksheet for your records.
Veterinary Portable Multi-parameter Patient Monitor. Operation Manual
 Veterinary Portable Multi-parameter Patient Monitor Operation Manual Content Chapter 1 Introduction... 1-1 1.1 General Information... 1-3 1.2 Screen Display... 1-4 1.3 Button Functions and Basic Operation...
Veterinary Portable Multi-parameter Patient Monitor Operation Manual Content Chapter 1 Introduction... 1-1 1.1 General Information... 1-3 1.2 Screen Display... 1-4 1.3 Button Functions and Basic Operation...
SERVICE MANUAL YM6000 Patient Monitor
 SERVICE MANUAL YM6000 Patient Monitor EU representative TECNOMED 2000 S.L. Valencia, 25 28012 Madrid Spain Manufacturer Mediana Co., Ltd. Wonju Medical Industry Park, 1650 1 Donghwa ri, Munmak eup, Wonju
SERVICE MANUAL YM6000 Patient Monitor EU representative TECNOMED 2000 S.L. Valencia, 25 28012 Madrid Spain Manufacturer Mediana Co., Ltd. Wonju Medical Industry Park, 1650 1 Donghwa ri, Munmak eup, Wonju
The information in this document is subject to change without notice.
 Operator s Manual NOTICE The information in this document is subject to change without notice. Welch Allyn makes no warranty of any kind on this material, including but not limited to, the implied warranties
Operator s Manual NOTICE The information in this document is subject to change without notice. Welch Allyn makes no warranty of any kind on this material, including but not limited to, the implied warranties
Australian/New Zealand Standard
 AS/NZS 3200.2.4:2006 IEC 60601-2-4:2005 AS/NZS 3200.2.4:2006 Australian/New Zealand Standard Medical electrical equipment Part 2.4: Particular requirements for safety Cardiac defibrillators AS/NZS 3200.2.4:2006
AS/NZS 3200.2.4:2006 IEC 60601-2-4:2005 AS/NZS 3200.2.4:2006 Australian/New Zealand Standard Medical electrical equipment Part 2.4: Particular requirements for safety Cardiac defibrillators AS/NZS 3200.2.4:2006
User s Reference Manual Volume 1 System Information
 Philips M1165/66/67/75/76/77A CMS Patient Monitoring System and Philips M1205A V24 and V26 Patient Monitor User s Reference Manual Volume 1 System Information PHI Part Number M1046-9220L Printed 02/2003
Philips M1165/66/67/75/76/77A CMS Patient Monitoring System and Philips M1205A V24 and V26 Patient Monitor User s Reference Manual Volume 1 System Information PHI Part Number M1046-9220L Printed 02/2003
Product Specifications
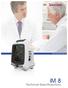 1.Safety Specifications 1.1Product Classification Class Type of protection against electric shock Specification Class I, with internal power supply Degree of protection against electric shock Anti-defibrillation:
1.Safety Specifications 1.1Product Classification Class Type of protection against electric shock Specification Class I, with internal power supply Degree of protection against electric shock Anti-defibrillation:
B40/B20 Patient Monitor User's Guide. English E (Paper) 21 November General Electric Company. All rights reserved.
 B40/B20 Patient Monitor User's Guide English 2081499-002 E (Paper) 21 November 2014 2014 General Electric Company. All rights reserved. B40/B20 Patient Monitor User's Guide Related to software VSP-C 0459
B40/B20 Patient Monitor User's Guide English 2081499-002 E (Paper) 21 November 2014 2014 General Electric Company. All rights reserved. B40/B20 Patient Monitor User's Guide Related to software VSP-C 0459
SERVICE MANUAL. Patient Monitor M30. EU representative TECNOMED 2000 S.L. Valencia, Madrid Spain
 SERVICE MANUAL Patient Monitor M30 EU representative TECNOMED 2000 S.L. Valencia, 25-28012 Madrid Spain Manufacturer Mediana Co., Ltd. Wonju Medical Industry Park, 1650-1 Donghwa-ri, Munmak-eup, Wonju-si,
SERVICE MANUAL Patient Monitor M30 EU representative TECNOMED 2000 S.L. Valencia, 25-28012 Madrid Spain Manufacturer Mediana Co., Ltd. Wonju Medical Industry Park, 1650-1 Donghwa-ri, Munmak-eup, Wonju-si,
Track the truth. Give no false assurance. Do no harm. Nellcor SpO 2. Module for use with Philips IntelliVue * Patient Monitors
 Track the truth Give no false assurance Do no harm Nellcor Module for use with Philips IntelliVue * Patient Monitors Nellcor Module Features Designed to be used in a range of critical care environments
Track the truth Give no false assurance Do no harm Nellcor Module for use with Philips IntelliVue * Patient Monitors Nellcor Module Features Designed to be used in a range of critical care environments
M9500 Patient Monitor User s Manual
 M9500 Patient Monitor User s Manual Guangdong Biolight Meditech Co., Ltd. Address: Innovation First Road, Technology Innovation Coast, Jinding, Zhuhai, P.R.CHINA Tel: +86-756-3399900 Fax: +86-756-3399989
M9500 Patient Monitor User s Manual Guangdong Biolight Meditech Co., Ltd. Address: Innovation First Road, Technology Innovation Coast, Jinding, Zhuhai, P.R.CHINA Tel: +86-756-3399900 Fax: +86-756-3399989
DESIGN ENGINEERING MANUFACTURING
 www.biometriccables.com QUALITY SAFETY RELIABILITY CUSTOM SOLUTIONS A28, Phase 3, SIDCO Industrial Estate Guindy, Chennai - 600 032 T: +91 44 22500953, 22500954 info@biometriccables.com DESIGN ENGINEERING
www.biometriccables.com QUALITY SAFETY RELIABILITY CUSTOM SOLUTIONS A28, Phase 3, SIDCO Industrial Estate Guindy, Chennai - 600 032 T: +91 44 22500953, 22500954 info@biometriccables.com DESIGN ENGINEERING
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 80601-2-55 First edition 2011-12-15 Medical electrical equipment Part 2-55: Particular requirements for the basic safety and essential performance of respiratory gas monitors
INTERNATIONAL STANDARD ISO 80601-2-55 First edition 2011-12-15 Medical electrical equipment Part 2-55: Particular requirements for the basic safety and essential performance of respiratory gas monitors
Alarm Fatigue in the ICU. Lynn Maguire MS, RN, PMC, NE-BC
 Alarm Fatigue in the ICU Lynn Maguire MS, RN, PMC, NE-BC Objectives At the completion of this lecture the participant will: Define Alarm Fatigue Discuss the Joint Commission s Patient Safety Goal regarding
Alarm Fatigue in the ICU Lynn Maguire MS, RN, PMC, NE-BC Objectives At the completion of this lecture the participant will: Define Alarm Fatigue Discuss the Joint Commission s Patient Safety Goal regarding
Propaq LT Outline Train the Trainer
 Train the Trainer A. Propaq LT and LTR Propaq LT Standalone monitor Propaq LTR Monitor that has the wireless Acuity feature enabled 1. Lightweight (Less than 2 pounds) 2. Uses rechargeable lithium ion
Train the Trainer A. Propaq LT and LTR Propaq LT Standalone monitor Propaq LTR Monitor that has the wireless Acuity feature enabled 1. Lightweight (Less than 2 pounds) 2. Uses rechargeable lithium ion
OPERATOR S MANUAL. NPB-4000/4000C Patient Monitor
 OPERATOR S MANUAL NPB-4000/4000C Patient Monitor Nellcor Puritan Bennett Inc. is an affiliate of Tyco Healthcare. Nellcor, Nellcor Puritan Bennett, Durasensor, C-LOCK, Oxisensor II, Dura-Y, and the Nellcor
OPERATOR S MANUAL NPB-4000/4000C Patient Monitor Nellcor Puritan Bennett Inc. is an affiliate of Tyco Healthcare. Nellcor, Nellcor Puritan Bennett, Durasensor, C-LOCK, Oxisensor II, Dura-Y, and the Nellcor
DPM 4 Patient Monitor. Operator s Manual
 DPM 4 Patient Monitor Operator s Manual I Intellectual Property Statement Mindray DS USA, Inc. (hereinafter called Mindray DS) owns the intellectual property rights to this product and this manual. This
DPM 4 Patient Monitor Operator s Manual I Intellectual Property Statement Mindray DS USA, Inc. (hereinafter called Mindray DS) owns the intellectual property rights to this product and this manual. This
Patient Monitoring System YM5500. Service Manual
 Patient Monitoring System YM5500 Service Manual SERVICE MANUAL For YM5500 Patient Monitor Mediana Co.,Ltd. 231-6 Nonhyun-Dong, Kangnam-Ku, Seoul, Korea Tel: (82) 2 542 3375 Fax: (82) 2 542 7447 YM5500
Patient Monitoring System YM5500 Service Manual SERVICE MANUAL For YM5500 Patient Monitor Mediana Co.,Ltd. 231-6 Nonhyun-Dong, Kangnam-Ku, Seoul, Korea Tel: (82) 2 542 3375 Fax: (82) 2 542 7447 YM5500
13799 Commerce Parkway Richmond, British Columbia V6V 2N9 Canada Benjamin Barker, Senior Manager Accreditations
 IAS Accreditation Number Company Name Address Contact Name Telephone (604) 273-4581 Effective Date of Scope February 9, 2018 Accreditation Standard ISO/IEC 17025:2005 TL-270 - Vancouver 13799 Commerce
IAS Accreditation Number Company Name Address Contact Name Telephone (604) 273-4581 Effective Date of Scope February 9, 2018 Accreditation Standard ISO/IEC 17025:2005 TL-270 - Vancouver 13799 Commerce
PM-9000Vet Veterinary Portable Multi-Parameter Patient Monitor. Operator s Manual
 PM-9000Vet Veterinary Portable Multi-Parameter Patient Monitor Operator s Manual CE Marking The product bears CE mark indicating its conformity with the provisions of the Council Directive 84/539/EEC
PM-9000Vet Veterinary Portable Multi-Parameter Patient Monitor Operator s Manual CE Marking The product bears CE mark indicating its conformity with the provisions of the Council Directive 84/539/EEC
SECTION L MONITORING EQUIPMENT
 BREATH-ALERT Y1090 BREATH-ALERT MK4 MONITOR The Breath-Alert Mk4 respiratory monitor is a simple yet sensitive and reliable device for monitoring breathing. A heat-sensitive sensor is placed in the breath
BREATH-ALERT Y1090 BREATH-ALERT MK4 MONITOR The Breath-Alert Mk4 respiratory monitor is a simple yet sensitive and reliable device for monitoring breathing. A heat-sensitive sensor is placed in the breath
13799 Commerce Parkway Richmond, British Columbia V6V 2N9 Canada Benjamin Barker, Manager Accreditations
 IAS Accreditation Number Company Name Address Contact Name Telephone (416) 747-4013 Effective Date of Scope May 22, 2017 Accreditation Standard ISO/IEC 17025:2005 TL-270 13799 Commerce Parkway Richmond,
IAS Accreditation Number Company Name Address Contact Name Telephone (416) 747-4013 Effective Date of Scope May 22, 2017 Accreditation Standard ISO/IEC 17025:2005 TL-270 13799 Commerce Parkway Richmond,
Alarm Management at Boston Medical Center A Roadmap to Safe Silence
 Alarm Management at Boston Medical Center A Roadmap to Safe Silence Jim Piepenbrink, Director Department of Clinical Engineering Co-Chair, Clinical Alarm Task Force This presentation is a view into the
Alarm Management at Boston Medical Center A Roadmap to Safe Silence Jim Piepenbrink, Director Department of Clinical Engineering Co-Chair, Clinical Alarm Task Force This presentation is a view into the
Welch Allyn Connex Devices
 Welch Allyn Connex Devices Directions for use Software version 2.4X 2018 Welch Allyn. All rights are reserved. To support the intended use of the product described in this publication, the purchaser of
Welch Allyn Connex Devices Directions for use Software version 2.4X 2018 Welch Allyn. All rights are reserved. To support the intended use of the product described in this publication, the purchaser of
EUROPEAN COMMITTEE FOR ELECTROTECHNICAL STANDARDIZATION (CENELEC) TECHNICAL COMMITTEE No. 62: ELECTRICALEQUIPMENT IN MEDICAL PRACTICE
 TC62/SEC1015/INF October 2009 EUROPEAN COMMITTEE FOR ELECTROTECHNICAL STANDARDIZATION (CENELEC) TECHNICAL COMMITTEE No. 62: ELECTRICALEQUIPMENT IN MEDICAL PRACTICE SOFTWARE AND MEDICAL DEVICES WORKING
TC62/SEC1015/INF October 2009 EUROPEAN COMMITTEE FOR ELECTROTECHNICAL STANDARDIZATION (CENELEC) TECHNICAL COMMITTEE No. 62: ELECTRICALEQUIPMENT IN MEDICAL PRACTICE SOFTWARE AND MEDICAL DEVICES WORKING
SureSigns VS4. Vital Signs Monitor Technical Data Sheet
 SureSigns VS4 Vital Signs Monitor Technical Data Sheet The SureSigns VS4 is a vital signs monitor that measures blood pressure, pulse rate, oxygen saturation (SpO 2 ), and temperature. The VS4 monitor
SureSigns VS4 Vital Signs Monitor Technical Data Sheet The SureSigns VS4 is a vital signs monitor that measures blood pressure, pulse rate, oxygen saturation (SpO 2 ), and temperature. The VS4 monitor
OPERATOR S MANUAL. Patient Monitor M20. EU representative TECNOMED 2000 S.L. Valencia, Madrid Spain
 OPERATOR S MANUAL Patient Monitor M20 EU representative TECNOMED 2000 S.L. Valencia, 25-28012 Madrid Spain Manufacturer Mediana Co., Ltd. Wonju Medical Industry Park, 1650-1 Donghwa-ri, Munmak-eup, Wonju-si,
OPERATOR S MANUAL Patient Monitor M20 EU representative TECNOMED 2000 S.L. Valencia, 25-28012 Madrid Spain Manufacturer Mediana Co., Ltd. Wonju Medical Industry Park, 1650-1 Donghwa-ri, Munmak-eup, Wonju-si,
SAINT JAMES HOSPITAL GROUP PURCHASING DEPARTMENT
 SAINT JAMES HOSPITAL GROUP PURCHASING DEPARTMENT CALL FOR QUOTES Q0001/SJH/01/11 Purchasing Department, George Borg Olivier Street, Sliema. SLM 1807. Tel: +356 23 292009 / +356 21 335235 Fax: +356 23 000000
SAINT JAMES HOSPITAL GROUP PURCHASING DEPARTMENT CALL FOR QUOTES Q0001/SJH/01/11 Purchasing Department, George Borg Olivier Street, Sliema. SLM 1807. Tel: +356 23 292009 / +356 21 335235 Fax: +356 23 000000
[Docket No. FAA ; Product Identifier 2017-NE-02-AD; Amendment ; AD ]
![[Docket No. FAA ; Product Identifier 2017-NE-02-AD; Amendment ; AD ] [Docket No. FAA ; Product Identifier 2017-NE-02-AD; Amendment ; AD ]](/thumbs/84/90584561.jpg) [Federal Register Volume 82, Number 189 (Monday, October 2, 2017)] [Rules and Regulations] [Pages 45710-45712] From the Federal Register Online via the Government Publishing Office [www.gpo.gov] [FR Doc
[Federal Register Volume 82, Number 189 (Monday, October 2, 2017)] [Rules and Regulations] [Pages 45710-45712] From the Federal Register Online via the Government Publishing Office [www.gpo.gov] [FR Doc
INSTRUCTIONS FOR USE. IntelliVue X2. Multi-Measurement Module Release G.0 with Software Revision G.0x.xx. Patient Monitoring
 INSTRUCTIONS FOR USE IntelliVue X2 Multi-Measurement Module Release G.0 with Software Revision G.0x.xx Patient Monitoring Printed in Germany 09/08 *M3002-9001B* Part Number M3002-9001B 4512 610 28261 S
INSTRUCTIONS FOR USE IntelliVue X2 Multi-Measurement Module Release G.0 with Software Revision G.0x.xx Patient Monitoring Printed in Germany 09/08 *M3002-9001B* Part Number M3002-9001B 4512 610 28261 S
Multiparameter. er Monitor DM
 d e n t a l a n d m e d i c a l e q u i p m e n t User manual Multiparameter er Monitor DM ENGLISH Contents 1 Introduction...1 1.1 General Information...1 1.2 Screen Display...2 1.3 Button Functions...5
d e n t a l a n d m e d i c a l e q u i p m e n t User manual Multiparameter er Monitor DM ENGLISH Contents 1 Introduction...1 1.1 General Information...1 1.2 Screen Display...2 1.3 Button Functions...5
Alarm Management. Objectives
 Alarm Management Michelle Bell, RN, BSN, FISMP, CPPS Kelly Graham, BS, RN Objectives 1. Discuss the importance of appropriate alarm use 2. Describe strategies for improving alarm safety and how to begin
Alarm Management Michelle Bell, RN, BSN, FISMP, CPPS Kelly Graham, BS, RN Objectives 1. Discuss the importance of appropriate alarm use 2. Describe strategies for improving alarm safety and how to begin
İndex SUCTION UNIT... PATIENT MONITOR HOLTER SYSTEM... 7
 PRODUCT CATALOGUE 1 2 İndex SUCTION UNIT... 4 PATIENT MONITOR... 5-6 HOLTER SYSTEM... Tourniquet Systems... Electrosurgical Units... Fetal MONITORS... Larygoscope Sets... Diagnostic Sets... - - 12 3 Suction
PRODUCT CATALOGUE 1 2 İndex SUCTION UNIT... 4 PATIENT MONITOR... 5-6 HOLTER SYSTEM... Tourniquet Systems... Electrosurgical Units... Fetal MONITORS... Larygoscope Sets... Diagnostic Sets... - - 12 3 Suction
PULSE OXIMETER PACKAGE
 instrumentation and software for research PULSE OXIMETER PACKAGE CANL-425SV-A USER S MANUAL DOC-095 Rev. 1.6 Copyright 2012 All Rights Reserved Med Associates Inc. P.O. Box 319 St. Albans, Vermont 05478
instrumentation and software for research PULSE OXIMETER PACKAGE CANL-425SV-A USER S MANUAL DOC-095 Rev. 1.6 Copyright 2012 All Rights Reserved Med Associates Inc. P.O. Box 319 St. Albans, Vermont 05478
Manufacturer s Responsibility Only under the following circumstances will manufacturer be responsible for the safety, reliability and performance of t
 Preface Thank you for using M9500 patient monitor. In order to enable you to skillfully operate Monitor as soon as possible, we provide this user s manual with delivery. When you install and use this instrument
Preface Thank you for using M9500 patient monitor. In order to enable you to skillfully operate Monitor as soon as possible, we provide this user s manual with delivery. When you install and use this instrument
Infinity M300 Patient Worn Monitoring
 Infinity M300 Patient Worn Monitoring Managing the care of ambulatory patients is challenging because you need to balance mobility with patient safety. The innovative Infinity M300 patientworn monitor
Infinity M300 Patient Worn Monitoring Managing the care of ambulatory patients is challenging because you need to balance mobility with patient safety. The innovative Infinity M300 patientworn monitor
1 Appendix - Product Specifications
 1 Appendix - Product Specifications WARNING The patient monitor may not meet its performance specification if stored or used outside the manufacturer s specified temperature and humidity range. 1.1 Safety
1 Appendix - Product Specifications WARNING The patient monitor may not meet its performance specification if stored or used outside the manufacturer s specified temperature and humidity range. 1.1 Safety
New DATE OF BLDG. PERMIT OR PLAN APPROVAL: SURVEYOR (SIGNATURE) TITLE OFFICE DATE REVIEW AUTHORITY OFFICIAL (SIGNATURE) TITLE OFFICE DATE
 DEPARTMENT OF HEALTH & HUMAN SERVICES CENTERS FOR MEDICARE & MEDICAID SERVICES FIRE SAFETY SURVEY REPORT - AMBULATORY SURGICAL CENTERS Medicare 1. (A) PROVIDER NUMBER Identifying information as shown in
DEPARTMENT OF HEALTH & HUMAN SERVICES CENTERS FOR MEDICARE & MEDICAID SERVICES FIRE SAFETY SURVEY REPORT - AMBULATORY SURGICAL CENTERS Medicare 1. (A) PROVIDER NUMBER Identifying information as shown in
Tesla M3. MRI Patient Monitoring System. Instructions for Use. Version 4.0
 Instructions for Use Version 4.0 Tesla M3 MRI Patient Monitoring System 0120 1 MIPM Mammendorfer Institut für Physik und Medizin GmbH, herein after called MIPM. Printed in Germany Subject to change without
Instructions for Use Version 4.0 Tesla M3 MRI Patient Monitoring System 0120 1 MIPM Mammendorfer Institut für Physik und Medizin GmbH, herein after called MIPM. Printed in Germany Subject to change without
<< COMPANY LETTERHEAD >>
 Healthcare Facilities Accreditation Program Attn: Engineering Department 142 E. Ontario Street Chicago, IL 60611 > < Date > RE: Request for Code Equivalency ABC General Hospital,
Healthcare Facilities Accreditation Program Attn: Engineering Department 142 E. Ontario Street Chicago, IL 60611 > < Date > RE: Request for Code Equivalency ABC General Hospital,
740 SELECT. Multi-Parameter Monitor
 Multi-Parameter Monitor User Manual Addendum - CO2 Parameters This User Manual Addendum describes the features and operations of the Multi-Parameter monitor: Software Version 2.2 or above. 21-22-0333 Rev
Multi-Parameter Monitor User Manual Addendum - CO2 Parameters This User Manual Addendum describes the features and operations of the Multi-Parameter monitor: Software Version 2.2 or above. 21-22-0333 Rev
URGENT - Field Safety Notice Medical Device Correction
 Dear Customer, According to our records, you currently have one of the V60 ventilators noted above. This Field Safety Notice (FSN) is to advise you that Respironics California, LLC ( Respironics ) is voluntarily
Dear Customer, According to our records, you currently have one of the V60 ventilators noted above. This Field Safety Notice (FSN) is to advise you that Respironics California, LLC ( Respironics ) is voluntarily
LETTERS FOR EUROPEAN UNION MEDDEV RT021
 LETTERS FOR EUROPEAN UNION MEDDEV RT021 1.1. First Consignee (Distributor/Retailer) Urgent FIELD SAFETY NOTICE Letter (RT021) [Date] Fisher & Paykel Healthcare Limited 15 Maurice Paykel Place, East Tamaki
LETTERS FOR EUROPEAN UNION MEDDEV RT021 1.1. First Consignee (Distributor/Retailer) Urgent FIELD SAFETY NOTICE Letter (RT021) [Date] Fisher & Paykel Healthcare Limited 15 Maurice Paykel Place, East Tamaki
PENALTIES. All imported products are required to meet the same standards as domestic goods. National Safety Requirements. Customs Requirements
 1 of 15 Short Course On: Importation Requirements of Laser Products ICALEO 2006 Version 11-01-06wbkgrd Course Topics Discuss the penalties associated with improperly or illegally importing a laser product.
1 of 15 Short Course On: Importation Requirements of Laser Products ICALEO 2006 Version 11-01-06wbkgrd Course Topics Discuss the penalties associated with improperly or illegally importing a laser product.
This is a preview - click here to buy the full publication
 TECHNICAL REPORT IEC TR 60601-4-2 Edition 1.0 2016-05 colour inside Medical electrical equipment Part 4-2: Guidance and interpretation Electromagnetic immunity: performance of medical electrical equipment
TECHNICAL REPORT IEC TR 60601-4-2 Edition 1.0 2016-05 colour inside Medical electrical equipment Part 4-2: Guidance and interpretation Electromagnetic immunity: performance of medical electrical equipment
Spacelabs Healthcare - Connecting Innovation with Care
 Ultraview SL3800 Spacelabs Healthcare - Connecting Innovation with Care Spacelabs Healthcare is fully committed to making strong connections: between clinicians and their patients; between technological
Ultraview SL3800 Spacelabs Healthcare - Connecting Innovation with Care Spacelabs Healthcare is fully committed to making strong connections: between clinicians and their patients; between technological
PM Portable Multi-parameter Patient Monitor. Operation Manual
 PM-8000 Portable Multi-parameter Patient Monitor Operation Manual Intellectual Property Statement SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD. (hereinafter called Mindray) owns the intellectual
PM-8000 Portable Multi-parameter Patient Monitor Operation Manual Intellectual Property Statement SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD. (hereinafter called Mindray) owns the intellectual
LASER SAFETY TRAINING CATALOG LASER LASER INSTITUTE OF AMERICA S MAKE LASER SAFETY YOUR PRIORITY.
 MAKE LASER SAFETY YOUR PRIORITY LASER INSTITUTE OF AMERICA S LASER SAFETY TRAINING CATALOG www.lia.org 13501 Ingenuity Dr., Suite 128 32826 Phone: +1.407.380.1553 Fax: +1.407.380.5588 INDUSTRIAL/RESEARCH
MAKE LASER SAFETY YOUR PRIORITY LASER INSTITUTE OF AMERICA S LASER SAFETY TRAINING CATALOG www.lia.org 13501 Ingenuity Dr., Suite 128 32826 Phone: +1.407.380.1553 Fax: +1.407.380.5588 INDUSTRIAL/RESEARCH
Quick start guide. English
 Quick start guide English Read your entire Astral User Guide or Clinical Guide before use. The Astral device 1. Adapter port Can be fitted with single limb adapter, single limb leak adapter or double limb
Quick start guide English Read your entire Astral User Guide or Clinical Guide before use. The Astral device 1. Adapter port Can be fitted with single limb adapter, single limb leak adapter or double limb
URGENT: MEDICAL DEVICE CORRECTION Plum 360 Infusion System List Number: 30010
 . 600 N. Field Drive Lake Forest, IL 60045 (224) 212 2000 www.icumed.com 15 June 2017 URGENT: MEDICAL DEVICE CORRECTION Plum 360 Infusion System List Number: 30010 Dear Valued Plum 360 Infusion System
. 600 N. Field Drive Lake Forest, IL 60045 (224) 212 2000 www.icumed.com 15 June 2017 URGENT: MEDICAL DEVICE CORRECTION Plum 360 Infusion System List Number: 30010 Dear Valued Plum 360 Infusion System
Part 2-55: Medical electrical equipment. Particular requirements for the basic safety and essential performance of respiratory gas monitors
 This is a preview - click here to buy the full publication INTERNATIONAL STANDARD ISO 80601-2-55 Second edition 2018-02 Medical electrical equipment Part 2-55: Particular requirements for the basic safety
This is a preview - click here to buy the full publication INTERNATIONAL STANDARD ISO 80601-2-55 Second edition 2018-02 Medical electrical equipment Part 2-55: Particular requirements for the basic safety
Connex Integrated Wall System 2.2 Software Version
 Connex Integrated Wall System 2.2 Software Version Clinical Inservice Presentation MC12931 1 Table of Contents Device Overview & Intended Use Power ON Power OFF Home Screen Navigation Spot Profile Intervals
Connex Integrated Wall System 2.2 Software Version Clinical Inservice Presentation MC12931 1 Table of Contents Device Overview & Intended Use Power ON Power OFF Home Screen Navigation Spot Profile Intervals
Configuration Settings Appendix
 Configuration Settings Appendix Document Information: M2703-9401A Revision A.00.00 For monitor release C.0 with software revision C.00.xx. This document is an appendix to the Service Guide. Philips Medizin
Configuration Settings Appendix Document Information: M2703-9401A Revision A.00.00 For monitor release C.0 with software revision C.00.xx. This document is an appendix to the Service Guide. Philips Medizin
Atlas Monitor Outline Train the Trainer
 Atlas Monitor Outline Train the Trainer A. ATLAS Monitor 1. Atlas Monitor is not designed for use on children younger than 3 years old. 2. Weight: 9.5lbs to 13.2 lbs, depending on configuration 3. Variable
Atlas Monitor Outline Train the Trainer A. ATLAS Monitor 1. Atlas Monitor is not designed for use on children younger than 3 years old. 2. Weight: 9.5lbs to 13.2 lbs, depending on configuration 3. Variable
DEPARTMENT OF PUBLIC SAFETY DIVISION OF FIRE PREVENTION AND CONTROL
 DEPARTMENT OF PUBLIC SAFETY DIVISION OF FIRE PREVENTION AND CONTROL RULES AND REGULATIONS CONCERNING 8 CCR 1507-57 BUILDING AND FIRE CODE ENFORCEMENT AND CERTIFICATION OF INSPECTORS FOR LIMITED GAMING
DEPARTMENT OF PUBLIC SAFETY DIVISION OF FIRE PREVENTION AND CONTROL RULES AND REGULATIONS CONCERNING 8 CCR 1507-57 BUILDING AND FIRE CODE ENFORCEMENT AND CERTIFICATION OF INSPECTORS FOR LIMITED GAMING
Managing Clinical Alarms
 Managing Clinical Alarms Maria Cvach, DNP, RN, CCRN Assistant Director of Nursing, Clinical Standards Sharon Allan RN, MSN, ACNS BC, CCRC Clinical Nurse Specialist, CVSICU Matt Trojanowski, MSc, RRT Manager,
Managing Clinical Alarms Maria Cvach, DNP, RN, CCRN Assistant Director of Nursing, Clinical Standards Sharon Allan RN, MSN, ACNS BC, CCRC Clinical Nurse Specialist, CVSICU Matt Trojanowski, MSc, RRT Manager,
Propaq M Operator s Guide
 Propaq M Operator s Guide March 2011 9650-0803-01 Rev. B ZOLL is a registered trademark, and SurePower is a trademark of ZOLL Medical Corporation. Propaq and Smartcuf are registered trademarks, and SureBP
Propaq M Operator s Guide March 2011 9650-0803-01 Rev. B ZOLL is a registered trademark, and SurePower is a trademark of ZOLL Medical Corporation. Propaq and Smartcuf are registered trademarks, and SureBP
Capnostream 20p Bedside Capnography Monitor. The only integrated, complete picture of oxygenation and ventilation 1
 Capnostream 20p Bedside Capnography Monitor The only integrated, complete picture of oxygenation and ventilation 1 Smart Capnography and Pulse Oximetry Technology Smart Capnography is a suite of algorithms
Capnostream 20p Bedside Capnography Monitor The only integrated, complete picture of oxygenation and ventilation 1 Smart Capnography and Pulse Oximetry Technology Smart Capnography is a suite of algorithms
D Moving beyond telemetry INFINITY M300
 D-19757-2009 Moving beyond telemetry INFINITY M300 MT-4187-2006 D-19730-2009 MT-4488-2006 02 03 Transforming telemetry into patient-worn monitoring From the standpoint of patient monitoring, today s busy
D-19757-2009 Moving beyond telemetry INFINITY M300 MT-4187-2006 D-19730-2009 MT-4488-2006 02 03 Transforming telemetry into patient-worn monitoring From the standpoint of patient monitoring, today s busy
T1 Patient Monitor. Operator s Manual
 T1 Patient Monitor Operator s Manual Copyright 2014 Shenzhen Mindray Bio-Medical Electronics Co., Ltd. All rights reserved. For this Operator s Manual, the issue date is November 2014. WARNING Federal
T1 Patient Monitor Operator s Manual Copyright 2014 Shenzhen Mindray Bio-Medical Electronics Co., Ltd. All rights reserved. For this Operator s Manual, the issue date is November 2014. WARNING Federal
VS2000V Veterinary Vital Signs Monitor Operation Manual
 VS2000V Veterinary Vital Signs Monitor Operation Manual - English Version 1.0, July 2011 2011 UTECH Co., Ltd. All rights reserved. Table of Contents Table of Contents Warranty and Service Information...
VS2000V Veterinary Vital Signs Monitor Operation Manual - English Version 1.0, July 2011 2011 UTECH Co., Ltd. All rights reserved. Table of Contents Table of Contents Warranty and Service Information...
Instructor Guide. HeartStart MRx M3535A/M3536A
 Instructor Guide HeartStart MRx M3535A/M3536A Notice About This Edition Publication number 453564045041 Edition 6 Printed in the USA To determine the product level version to which this guide applies
Instructor Guide HeartStart MRx M3535A/M3536A Notice About This Edition Publication number 453564045041 Edition 6 Printed in the USA To determine the product level version to which this guide applies
PAT I E N T M O N I TO R I N G N E T W O R K
 Operating Instructions Datascope Panorama PAT I E N T M O N I TO R I N G N E T W O R K PAT I E N T M O N I TO R I N G N E T W O R K 0070-01-0656-01_revC.indd 1 3/10/10 5:26:09 PM Operating Instructions
Operating Instructions Datascope Panorama PAT I E N T M O N I TO R I N G N E T W O R K PAT I E N T M O N I TO R I N G N E T W O R K 0070-01-0656-01_revC.indd 1 3/10/10 5:26:09 PM Operating Instructions
T1 Monitor/Module Default Settings
 Hospital, City, ST Department Clinical/Sales/Service Representatives Date Software Version Signature,Title Maintenance System time Date format: Time format: 12 24 Manage Config Department: General Select
Hospital, City, ST Department Clinical/Sales/Service Representatives Date Software Version Signature,Title Maintenance System time Date format: Time format: 12 24 Manage Config Department: General Select
